Critical Care Reviews Meeting 2026
The countdown is on!
CCR26
The Best Critical Care Trials in the World
June 10th - 12th, 2026
Titanic Belfast, Belfast, Northern Ireland
iRehab
Results presentation at CCR Down Under 2025, Melbourne
TOWAR
The Last Drop
The REMAP CAP Ivermectin for COVID-19 Trial
Our latest blog is out
Some treatments save lives. Others teach medicine how to stop.
For centuries, bloodletting was medicine’s answer to illness: plausible, authoritative, visible, and wrong. It survived because it made sense — until clinicians began to count.
The Last Drop follows that old lesson into the COVID-19 pandemic, where ivermectin travelled from in-vitro signal to public certainty, influencer promotion, widespread use, and clinical demand before robust evidence caught up.
REMAP-CAP’s ivermectin trial did not produce a miracle. It produced something more uncomfortable, and perhaps just as important: evidence of non-benefit.
This is a story about bloodletting, ivermectin, REMAP-CAP, and why de-implementing a non-beneficial treatment can be as important as discovering a beneficial one.
added May 13th
CCR Trial Summaries & Critiques
CCR Meeting Talks
After 14 meetings, we have a huge collection of superb presentations. We'll showcase them here, one at a time.
CCR22 | FIRST-ABC Trial
Drs Padmanabhan Ramnarayan and Paul Mouncey (both London) present the results of the FIRST-ABC trial, comparing high-flow nasal cannulae therapy with CPAP in acute ill children with respiratory failure, at the Critical Care Reviews Meeting 2022. Dr Srinivas Murthy (Vancouver) delivers an editorial, which is followed by a panel discussion including Profs Kathryn Maitland (Kalifi), John Marshall (Toronto) and Marion Campbell (Aberdeen). The session is chaired by Prof Howard Bauchner.
added April 20th
FIRST-ABC (Step Down) Trial Presentation
Dr Padmanabhan Ramnarayan (London) and Mr Alvin Richards-Belle (London) present the results of the FIRST-ABC (Step Down) trial, comparing high flow nasal oxygen with continuous positive airway pressure in recently extubated children. This was presented during a Critical Care Reviews Livestream event.
Papers

RCT

RCT

RCT

Observational Study

Guideline

Guideline
added March 24th

Guideline
RCT
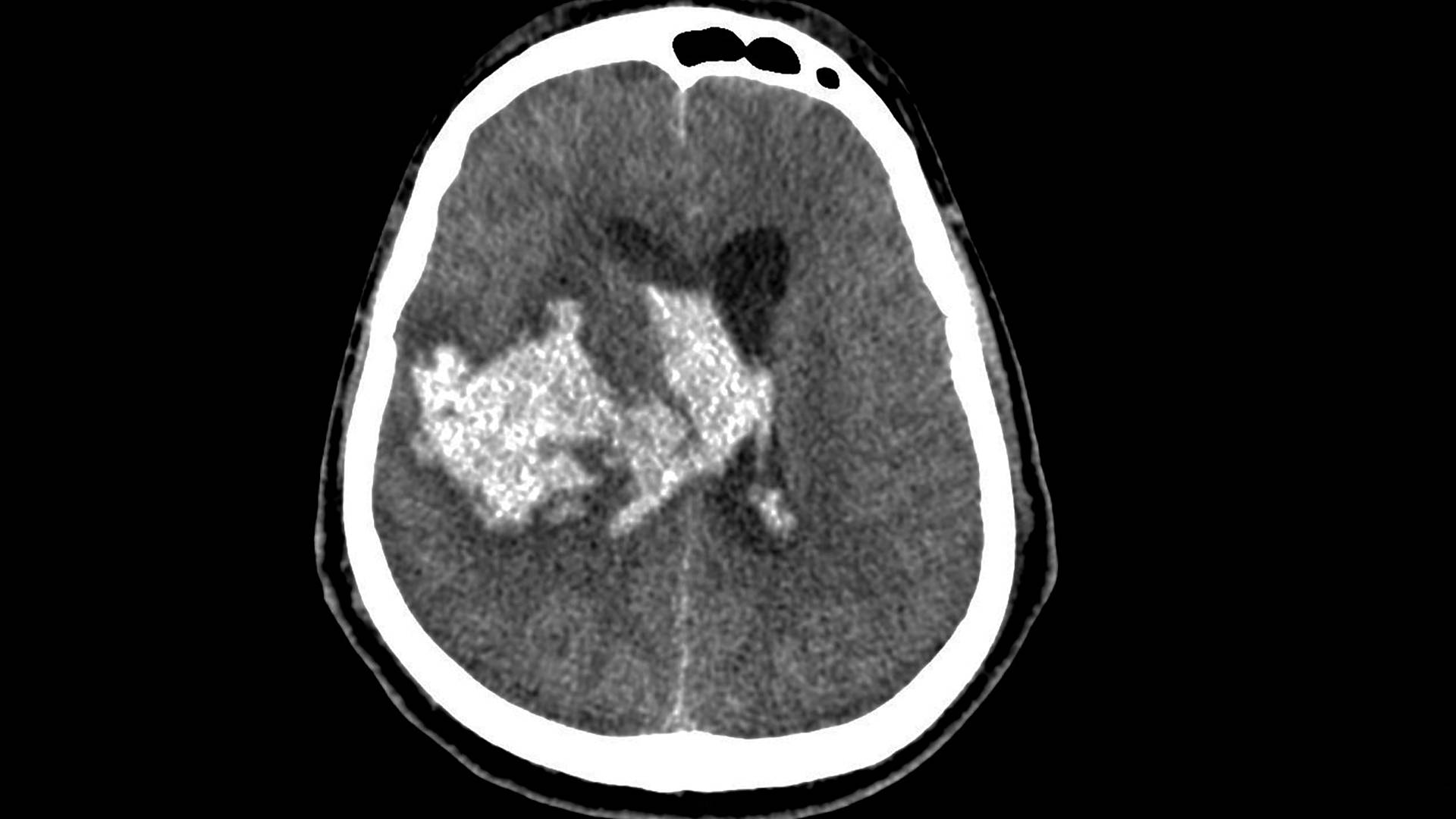
Commentary
added February 17th
Narrative Reviews
added February 17th
Foundational Trials Collection
After many months of preparation, we're delighted to release our Foundational Trials Collection.
250 critical care contemporary trials are summarised and critiqued in depth.
If you are new to the field, are undertaking exams, or just want to refresh your knowledge, this is an excellent place to start.
added February 17th

Sample Summaries
-
HEMOTION
-
ADRENAL
-
BLING III